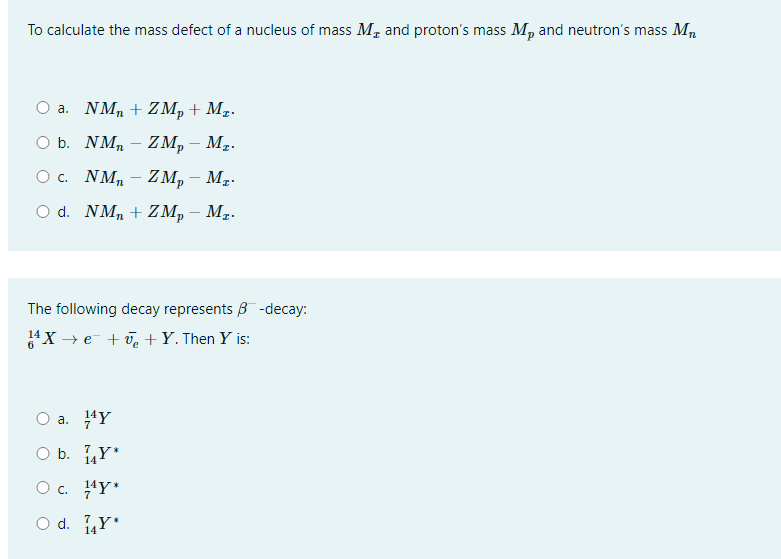
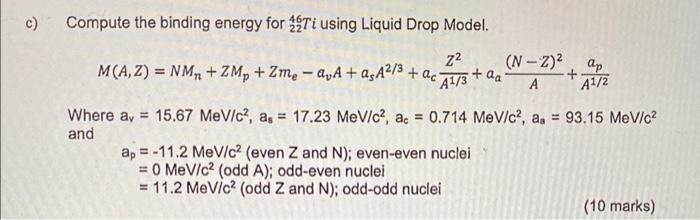If M(A, Z), Mp and Mn denote the masses of the nucleus, proton and neutron respectively in units of u(1u = 931.5 MeV/c^2) and BE represents its binding energy in MeV, then.

SOLVED: Semi-empirical mass formula: 2/3 22 (N-2)? B= #lap A 3/4 ahA-a,A a a, A1/3 ah =15.5 MeV a,=16.8 MeV 23 MeV ap = 34 MeV +1 1 = Zeven-N even nuclei

If M(A, Z), Mp and Mn denote the masses of the nucleus, proton and neutron respectively in units of u(1u = 931.5 MeV/c^2) and BE represents its binding energy in MeV, then.

The Nobel Prize on Twitter: "26-year-old Albert Einstein formulated the two postulates of special relativity on 30 June 1905 1. The Principle of Relativity 2. The Constancy of Speed of Light in

If M(A, Z), Mp and Mn denote the masses of the nucleus, proton and neutron respectively in units of u(1u = 931.5 MeV/c^2) and BE represents its binding energy in MeV, then.

Friends! I solved a small problem to determine the binding energy of nucleons in the nucleus of an atom. W… | Physics and mathematics, Binding energy, Physics books
![SOLVED: Question 2: [Marks: 4+6 ] (a) Explain as [0 why or why not the formula for total mass of a nucleus mw given below in term of proton and neutron masses SOLVED: Question 2: [Marks: 4+6 ] (a) Explain as [0 why or why not the formula for total mass of a nucleus mw given below in term of proton and neutron masses](https://cdn.numerade.com/ask_images/cdd84cc527c3438bb525e13980b1ebf3.jpg)
SOLVED: Question 2: [Marks: 4+6 ] (a) Explain as [0 why or why not the formula for total mass of a nucleus mw given below in term of proton and neutron masses

Mn and Mp represent mass of neutron and proton respectively. If an element having atomic mass M has N-neutron and Z-proton, then the correct relation will be :

SOLVED: The following masses are given: 1 n 1.008665 0 1 H 1.007825 u 1 226 Ra 226.025403 u 88 The binding energy of 226 Ra, in MeV, is closest to which value? 88 2500 2100 1900 2300 1700