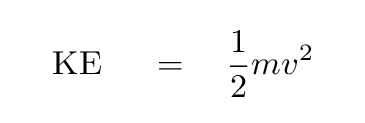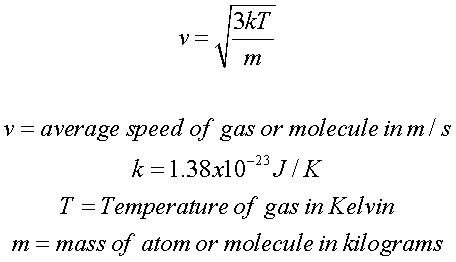a) Using the Bohr's model calculate the speed of the electron in a hydrogen atom in the n = 1, 2, and 3 levels. (b) Calculate the orbital period in each of these levels.
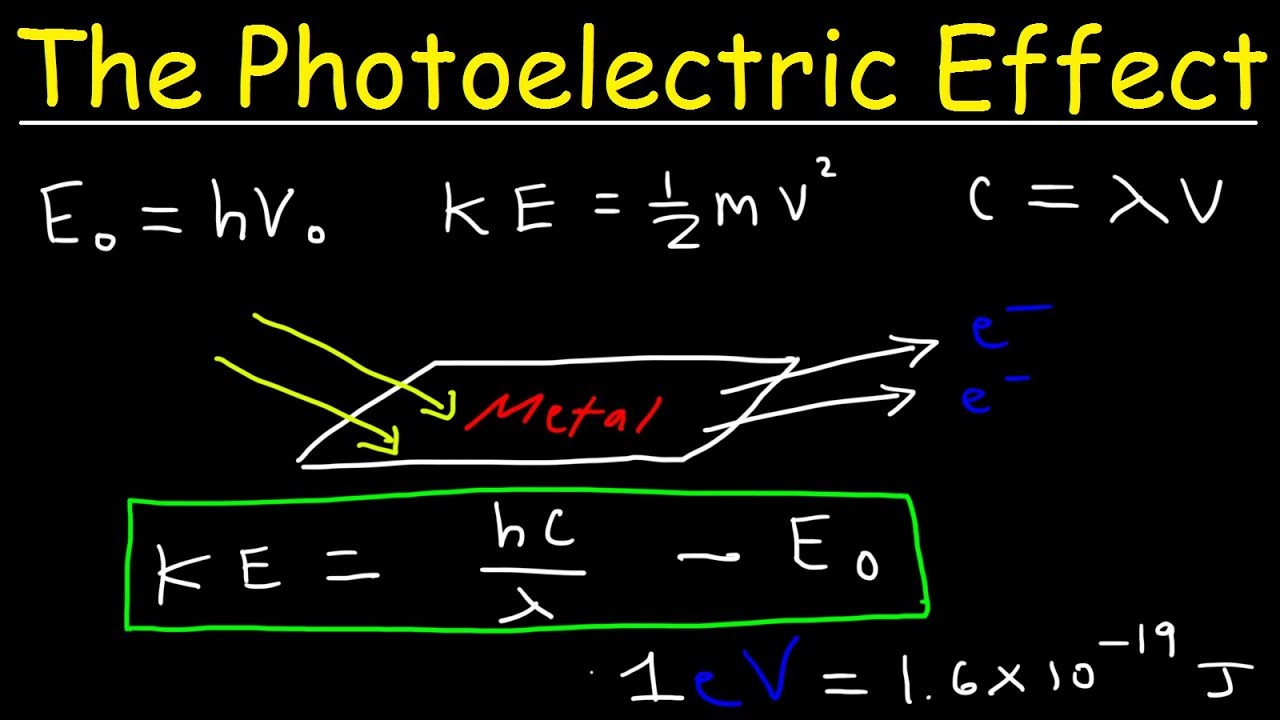
Photoelectric Effect, Work Function, Threshold Frequency, Wavelength, Speed & Kinetic Energy, Electr - YouTube
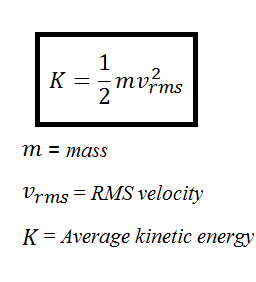
Kinetic Molecular Theory of Gases | Speed, Formula & Calculation - Video & Lesson Transcript | Study.com

How to Calculate the Average Kinetic Energy of Molecules in Gas at a Certain Temperature | Physics | Study.com
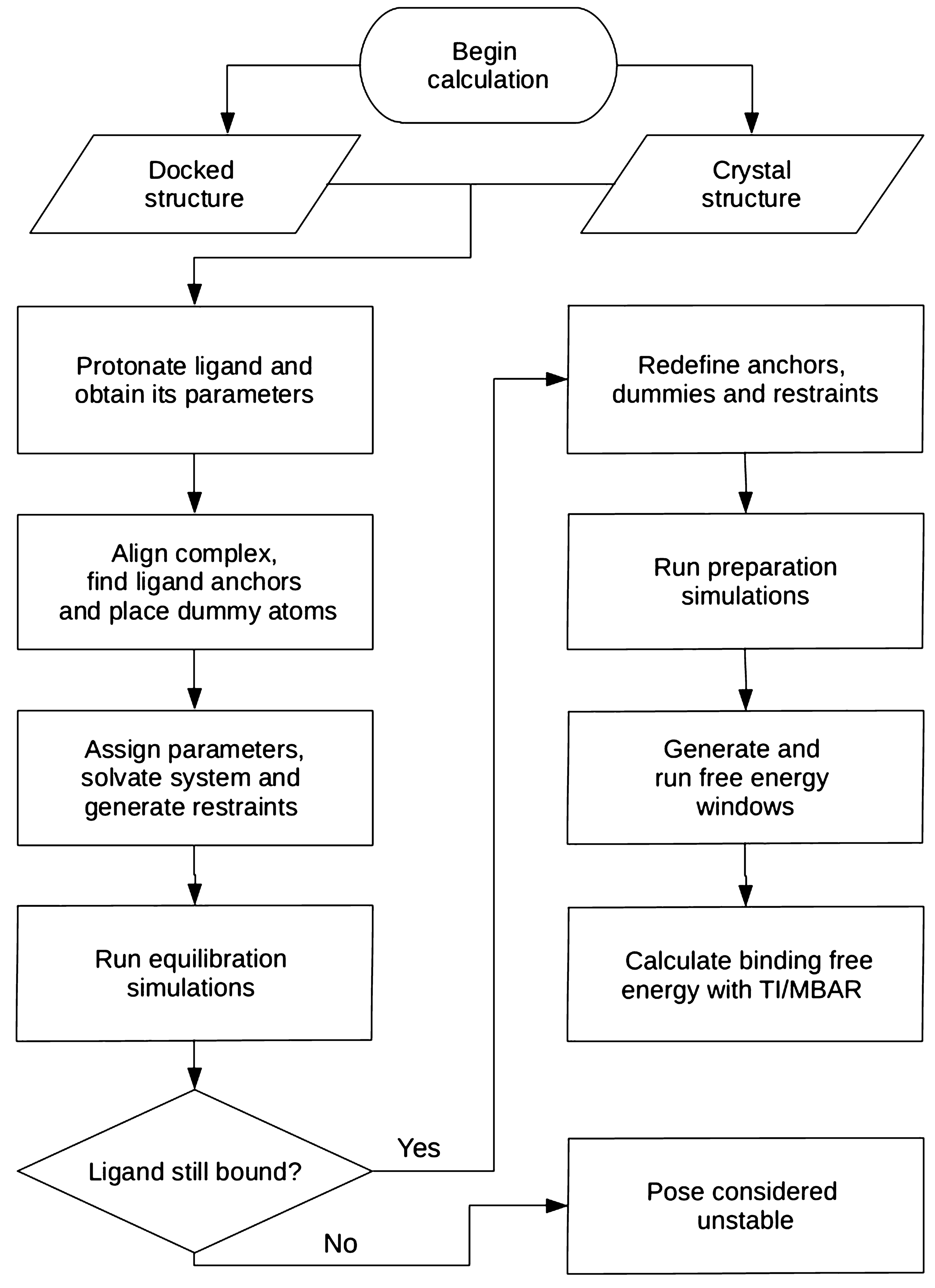
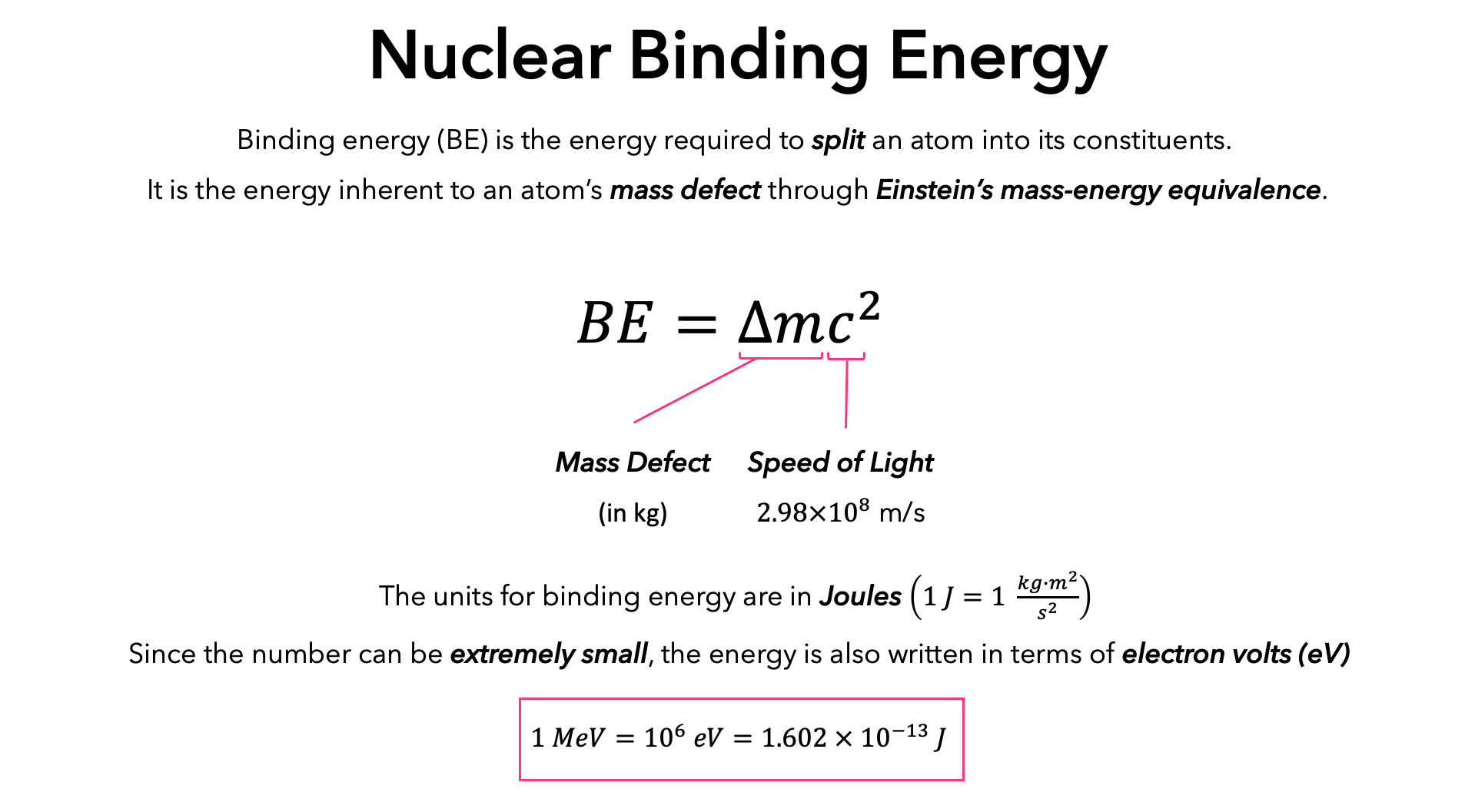



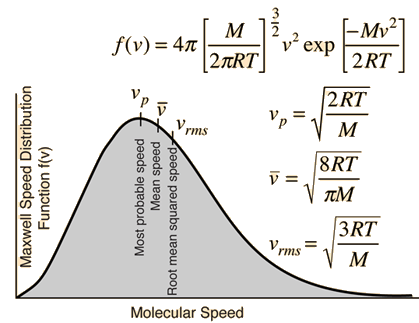


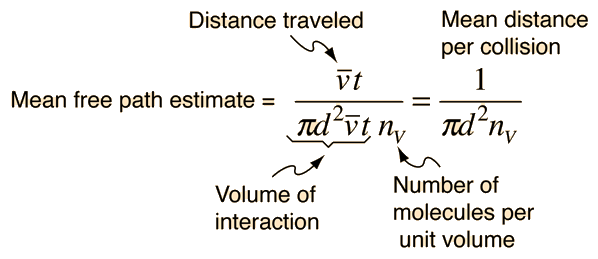
![Solved \[ K E=\frac{1}{2} m v^{2} \] amu \( | Chegg.com Solved \[ K E=\frac{1}{2} m v^{2} \] amu \( | Chegg.com](https://media.cheggcdn.com/media/a47/a47b7914-4da2-4219-9ba3-fff51b85fed0/phpCq4TTe)